You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
Sleep medicine is a young subspecialty. The medical community is becoming more aware of the significant role that sleep disturbances play in patients’ general health. Physicians who treat sleep disorders clearly understand the relationship between sleep and chronic pain, and they appreciate the key role that sleep plays in healing from both micro-trauma and macro-trauma. Many physicians have acknowledged a concern for the social problems associated with snoring. Most importantly, a greater understanding of the serious co-morbidities of hypertension, myocardial infarctions, stroke, weight gain, diabetes, acid reflux, and excessive daytime sleepiness leading to work and home injuries as well as motor vehicle accidents has made the treatment of sleep disorders a high priority. Medical professionals now understand that chronic sleep deprivation is related to a shortened life expectancy, and that quality sleep is potentially associated with improved overall mental and physical health.
Although there is still much about sleep physiology that remains a mystery, the science of sleep medicine has improved dramatically in recent years. While, like any new science, there is much to be learned, the recent awareness and improvements in therapy already have led to an improved quality of life for many patients. Yet, 90% of people with sleep-disordered breathing remain undiagnosed.1 The American Society of Anesthesiologists estimates that 16 million people have undiagnosed sleep disorders.2
The role that dentists can play in this critical field is extremely important.3,4 Many dentists tend to see their patients on a more regular basis than general physicians; consequently, it is very important that dentists learn to ask the proper questions so proper referrals can be made. In the best interest of patients’ needs, it is equally important that dentists avoid the pitfalls that can lead to improper therapy or a competitive environment with the very sleep physicians with whom they need to function in harmony. Providing treatment for patients who snore without formulating a complete and accurate diagnosis is problematic.
This approach, based solely on a patient’s report of snoring, can be compared with treating gingival hyperplasia without a proper diagnosis, including periodontal probing and radiographs. If the snoring is resolved, the patient often will not seek further therapy. Consequently, the obstructive disorder may continue and the patient remains at risk for hypertension, stroke, diabetes, and the other associated co-morbidities.5 Just as treating gingivitis without proper diagnosis would be considered below the standard of care, treating obstructive disorders carries the same diagnostic requirements. The consequences of sleep disturbances are potentially more severe than tooth loss from underlying periodontitis.6 In addition, any treatment of snoring without including the medical profession widens the gap between the professions, the same gap that needs to be narrowed to treat the vast majority of patients with obstructive sleep apnea appropriately.
NORMAL SLEEP STAGES
Normal sleep is divided into two types: non-REM (NREM) and REM.7-9 REM sleep is, of course, rapid eye movement. NREM sleep is divided into four distinct stages. Stages one through four are determined by alterations in brain wave activity as recorded in an electroencephalogram (EEG). Physiological changes occur with each sleep stage. During sleep, the body is considered to be in an inactive state while the brain remains active. A normal sleep pattern has four to five cycles throughout the night. NREM and REM sleep states alternate in cycles that usually last between 90 and 110 minutes. Normal NREM sleep occupies 75% of the night, and is characterized by a decrease in body temperature, blood pressure, breathing rate, and most physiologic functions.7 REM sleep is characterized by a highly active brain in an essentially paralyzed body.10,11
As a result of this pattern, normal sleep presents with specific relative time spent in each stage. Arousals (events that alter normal stage progression) cause alteration of this normal architecture and often are responsible for sleep disturbances and hypersomnolence, which is defined as excessive daytime sleepiness (Figure 1).7 Arousals can be caused by partial obstructions leading to snoring or difficult breathing, complete obstructions defined as apneas, non-obstructive conditions such as pain, central apneas (a stoppage of breathing from lack of respiratory effort as opposed to an obstruction), involuntary body movements, or even psychological situations.7 Of extreme importance to dentists, arousals can be associated with the parasomnia of bruxism.12-14
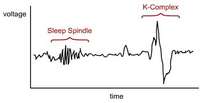
Consequently, parafunctional control has the potential to decrease the arousal index (the number of arousals per hour of sleep) and improve the quality of sleep. In NREM stage one, sleep usually occupies approximately 5% of the sleep cycle, and is represented by a relatively active brain-wave pattern.7 Stage two is represented by active brain waves that include specific EEG patterns called K-complexes and sleep spindles (Figure 2). This stage usually occupies 45% of the sleep cycle.7 Stages three and four combined are referred to as slow-wave sleep. Earlier in sleep medicine these stages often were referred to as Delta sleep.7 These stages represent progressively deeper sleep, are identified by slow high-amplitude brain waves, and usually occupy approximately 25% of the night’s pattern.7 These percentages do alter as patients age, with slow-wave sleep decreasing significantly in the sixth and seventh decades.7 Various medications as well as the existence of chronic pain also have effects on sleep architecture.15
REM sleep is characterized by rapid eye movement, irregular breathing, relative skeletal muscle paralysis, and an irregular heart rate. It is when most of the colorful and vivid dreaming occurs, and it occupies 20% to 25% of the normal sleep cycle.7 REM latency refers to the amount of time it takes from sleep onset to the first period of REM. Normal REM latency is about 90 minutes, and can be compromised with significant sleep debt or specific sleep disorders, such as narcolepsy.7-9
The gold standard for diagnosis of sleep disorders is the polysomnogram (PSG)16 (Figure 3). These overnight studies take place in sleep laboratories. Patients are monitored through the night. The PSG monitors brain waves (EEGs to determine sleep stages), eye movements (electrooculogram [EOG]), muscle tone (electromyography [EMG]), nasal air flow, blood oxygen levels, respirations, cardiac function, and respiratory effort. From these studies, physicians can determine if there is an alteration in patients’ normal sleep architecture. Patients are monitored throughout the night and often videotaped. The sleep studies then are scored, and among the plethora of information received and calculated is the respiratory disturbance index (RDI).17 The RDI represents the number of hypopneas (which has several definitions, the most common of which is a 3% drop in blood oxygen levels) plus apneas (universally described as total obstruction of air flow for 10 seconds or longer) per hour plus respiratory effort related arousals (RERAs) (drops in oxygen levels associated with arousals that do not meet the criteria of an hypopnea).7 EMGs on leg muscles are used to evaluate the existence of periodic leg movements. Recently, the placement of the surface EMG electrodes on the elevator musculature has allowed physicians to look more closely at parafunction (B.G. unpublished data).
CLASSIFICATION OF SLEEP DISORDERS
Sleep disorders are classified as dysomnias and parasomnias.Dysomnias are disorders of initiating and maintaining sleep, as well as disorders of excessive sleepiness. Examples of dysomnias are narcolepsy, insomnias, circadian rhythm disorders (eg, jet lag), and obstructive sleep disorders including sleep apnea.7 Parasomnias are undesirable physical phenomena that occur during sleep, such as sleep walking, nocturnal leg cramps, periodic leg movements, nightmares, and bruxism.7-9
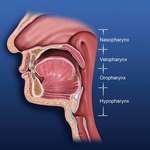
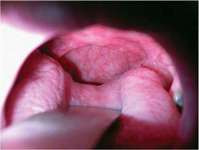
Obstructive breathing disorders fall on a continuum beginning with snoring, continuing with upper-airway resistance syndrome and hypopnea with associated hypoxemia, and ending with sleep apnea (Figure 4).An apnea is defined as the stoppage of breathing for 10 seconds or more.7 If the stoppage of breathing is caused by an obstruction in the airway, it is called an obstructive sleep apnea. Obstructive apneas are a result of the blockage of the airway, either at the junction of the oropharynx (soft palate and base of the tongue) or anywhere else in the pharyngeal anatomy (Figure 5).When the blockage occurs in the pharynx because of muscular collapse at that site, the pharynx is said to be compliant.
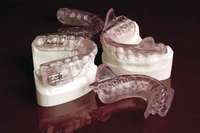
Most often obstructive apnea is a result of more than one site of obstruction.18 Central apnea refers to stoppage of breathing for 10 seconds or longer, not as a result of an obstruction but from the lack of respiratory effort.7 Often when there are central apneas, there are also obstructive apneas, and this condition is referred to as mixed apnea. For a diagnosis of central apnea to be made, 80% of the apnea events must occur centrally.7 Dentists can play a major role in the treatment of obstructive disorders. An understanding of these disorders, the method of action of oral appliances (Figure 6 through Figure 9), and the potential dental and non-dental side effects of these oral appliances will be the focus of the next section.
DENTAL SLEEP MEDICINE
Snoring is at the beginning of the continuum of sleep disorders. Snoring can be more than a social issue; not all snoring is benign. Therefore, all snoring patients need to be diagnosed as to whether they have a more complex obstructive disorder. As stated previously, treating snoring without a complete diagnosis is not advisable. An incomplete diagnosis could result in a missed opportunity to identify a more significant sleep-disordered breathing condition and possibly protect the patient from the life-threatening co-morbidities associated with obstructive sleep-disordered breathing.
It has been estimated that 90% of all patients with obstructive sleep disorders remain undiagnosed.19 The reasons are numerous. The best diagnosis can be made after a polysomnogram, a laboratory-controlled all-night sleep study that is admittedly cumbersome, not entirely reliable, and often not available.20 Clearly, alternative available testing procedures need to be considered.21 Family physicians often do not ask questions related to sleep, and family physicians do not see patients at the rate patients are seen by dentists. When asked why questions about sleep, such a critical aspect of health, are not included during examinations or interviews, family physicians refer to the limited amount of time they have for an examination as well as their frustration with the diagnostic procedures and treatment options.22 It is clear that the dentist’s role can be significant in helping to identify patients with sleep-disordered breathing, as well as to give the many patients who have difficulties using continuous positive airway pressure (CPAP) a potentially successful alternative. The dentist, therefore, can play a role in both diagnosis and treatment.
THE DENTIST’S ROLE IN DIAGNOSIS
No profession has been as successful as dentistry in the field of prevention. A patient is more likely to see his dentist on a regular basis than any other healthcare professional.23 It was long ago determined that dentists could play a major role in helping to uncover potential health risks in their patients. Medical updates on 6-month or yearly recall visits often include questions about basic health issues and blood pressure recordings. There is a movement in the dental and medical fields to educate dentists to ask basic questions about their patients’ quality of sleep.24 By simply asking if patients snore and if they wake up and feel refreshed, dentists can screen patients with additional questions (Table). Neck circumference alone has been shown to be an excellent predictor of apnea.25,26 Additional questions about excessive daytime sleepiness, the presence of witnessed apneas, or difficulty in falling asleep or staying asleep can be helpful. Further questioning related to many of the comorbidities associated with sleep-disordered breathing also may lead dentists to referral to the proper sleep specialist and/or other medical specialist. These questions should not only be related to hypertension and excessive thirst, but also about oral acidity and esophageal burning.27
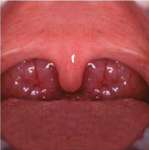
During the dental examination, dentists should look for excessive wear facets, which are likely to be a combination of parafunction and oral acidity. Obvious enamel defects from acid, including dentinal pooling (Figure 10), should be noted. Often the first signs of gastroesophageal reflux disease (GERD) are odontogenic, and there is a relationship between GERD and sleep apnea, with a proposed pathophysiology related to alterations in thoracic and abdominal pressure gradients.28,29 Positive responses should lead to referral to the sleep specialist. While the polysomnogram is indeed the gold standard in sleep medicine’s diagnostic regimen, various home-testing units are available (Figure 11).Many of these testing devices provide valuable information, yet they have some deficiencies as well.21,30 Most of them, at this point in time, do not evaluate EEG patterns and consequently do not detect alterations in sleep architecture. The lack of supervision with these home-testing units remains a concern; thus, at this time, these testing procedures generally are considered excellent screening devices but are not considered diagnostic by themselves and are generally not reimbursable by third-party payers in the
THE DENTIST’S ROLE IN TREATMENT
It is generally accepted that the gold standard of treatment for patients with sleep apnea is the use of CPAP.32 This effective therapy uses positive airway pressure to create a pneumatic air splint, reducing pharyngeal compliance. The amount of pressure required for each patient is determined in a titration procedure in a sleep laboratory. While it is a very effective therapy, patient compliance remains the most difficult obstacle to successful treatment. Despite recent improvements, which include a change from constant pressure to alternating pressures for inhalation and exhalation (called BiPAP), the addition of warm air humidifiers, and smaller, more portable, and quieter units, compliance after 6 months on many studies is well below 50%.33 Patients report problems with the fitting of the mask, air leakage, skin reactions to the air pressure, stomach bloating, claustrophobia, noisy machines, in-ability to initiate and maintain sleep, and total inconvenience.34-36
Options to CPAP use include either a surgical procedure or the use of an oral appliance. Surgery options were at one time very often performed. While the reported rate of success for the various surgical procedures vary, there is a significant level of pain associated with the most common procedure (uvulopalatalphyrgolplasty) and the most recent rates of success measured at 6 months are reported to be below 50%, with the percentage dropping over time (Figure 12).37 A laser-assisted procedure was very commonly used, but essentially has been abandoned because of its low level of success.38 Other procedures include bimaxillary orthognathic surgery and, in severe cases, tracheostomies. Both of these procedures tend to be very successful,but are,of course, not considered conservative therapies.39 Therefore, many careful considerations must be made before either procedure is recommended.
In 1995, a landmark study by Schmidt-Norwarwa and colleagues reported that the use of oral appliance therapy was effective for mild to moderate sleep apnea.40 A more recent study showed successful outcomes using this therapy for severe apnea in some cases.41 In addition, many sleep specialists consider the use of oral appliance therapy the treatment of choice for primary snoring and upper airway resistance syndrome.42,43 Many patients who use CPAP therapy successfully use oral appliance therapy when traveling or in combination with their CPAP device, which allows the low pressure to be more effective. When given the alternatives, many patients who do not comply with their CPAP therapy choose an oral appliance as their treatment of choice.44 The long-term compliance of oral appliance therapy is much higher than that of CPAP therapy.45-48 Clearly, oral appliance therapy plays a significant role in the treatment of patients with obstructive sleep disorders. In 2006, the American Academy of Sleep Medicine released their policy statement on oral appliances: Oral appliances are indicated in patients with mild to moderate [obstructive sleep apnea] who prefer their use to continuous positive airway pressure (CPAP) treatment or who are inappropriate candidates for or who are refractory to CPAP treatment.31 This statement does not suggest that oral appliance therapy should be the first line of therapy in all cases, nor does it suggest that it is inappropriate for severe cases.
The method of action of the oral appliance is well understood. By maintaining the mandible in a forward posture, the base of the tongue is pulled forward through the action of the genioglossus muscle (Figure 13). At the same time, action on the tensor veli palatine muscle is initiated through the attachments of the base of the tongue through the mandible and the ptergomandibular raphe. In addition, the alteration in the compliance of the pharynx with oral appliance therapy indicates a change in the tone of the pharyngeal musculature during sleep, including REM sleep, during which the musculature would normally be most compliant. Magnetic resonance imaging of the pharynx with the oral appliance in place has demonstrated an increase in the airway in the lateral dimension.49 Thus, the appliance is doing more than simply pulling the tongue forward and creating a decreased possibility of obstruction at the junction of the base of the tongue and the soft palate. Most often, obstructions occur at more than one site, and oral appliances remain potentially effective in these cases.
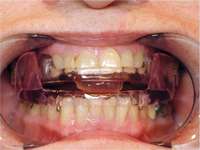
Specific training is required to treat patients with sleep-disordered breathing. Dentists should understand the nature of patients’ dysfunctions and work in a coordinated effort with sleep specialists. Dentists should discuss at length with their patients the possible side effects of oral appliance therapy, which include excessive salivation, temporomandibular joint pain, muscular pain and soreness, morning maladaptive mandibular posture, and occlusal changes (Figure 14).4
Treatment of these side effects involves joint and muscular therapy, the use of a leaf gauge on wakening as part of an occlusal maintenance program, and palliative therapeutic measures. Patient monitoring must be initiated with adaptation to the appliance, followed by guiding titration31 (Figure 15). The patient and dentist must work together and make well-advised risk vs benefit decisions during the treatment process. Too often there is a tendency for the dentist to be overly concerned about dental changes that may not affect function or esthetics, causing therapy to be abandoned when it is essential to the patient’s health. The treating dentist must be aware of the anatomy and physiology of the temporomandibular joint so that strains created in the joint can be recognized and treated as opposed to aborting oral appliance therapy, possibly prematurely.
CONCLUSION
Obstructive sleep disorders have the potential to negatively alter patients’ quality of life. Not only can the associated snoring be disruptive to the sleeping pattern of bed partners, but excessive daytime sleepiness, altered sleep architecture, and the co-morbidities of hypertension, cardiac disease, stroke, and GERD can debilitate patients further, decrease their quality of life, and significantly decrease their life expectancy. The vast majority of patients with sleep-disordered breathing are not diagnosed. Dentists have the potential to play a major role in both initiating diagnostic procedures and treating these patients. Dentists should not be cavalier and treat snoring, the first stage of this disease continuum, without a proper diagnosis. They must learn to work hand-in-hand with the medical community. Dentists must learn how to communicate on a regular basis and learn how to keep sleep physicians in the treatment loop. The treatment of sleep-disordered breathing is an art and a science, and to be successful, the dentist must be adept at both.
ACKNOWLEDGMENT
The author thanks Dr. Don Malizia for his indispensable research efforts.
REFERENCES
1. Baumel MJ, Maislin G, Pack AI. Population and occupational screening for obstructive sleep apnea: are we there yet? Am J Respir Crit Care Med. 1997;155(1):9-14.
2. Wake up to the effects of sleep apnea. 2008. American Society of Anesthesiologists Web site. Available at: http://www.asahq.org/patientEducation/apnea.htm. Accessed May 22, 2008.
3. Bailey DR, Attanasio R. Dentistry’s role in the management of sleep disorders. Recognition and management. Dent Clin North Am. 2001;45(4):619-630.
4. Schmidt-Nowara W. A review of sleep disorders. The history and diagnosis of sleep disorders related to the dentist. Dent Clin North Am. 2001;45(4):631-642.
5. Marklund M,
6. Lavie P, Lavie L, Herer P. All-cause mortality in males with sleep apnoea syndrome: declining mortality rates with age. Eur Respir J. 2005; 25(3):514-520.
7. Kryger MH, Roth T, Dement WC, eds. Principles and Practice of Sleep Medicine. 4th ed.
8. Silber MH, Krahn LE, Morgenthaler TI. Sleep Medicine in Clinical Practice.
9. Lavie P, Giora P, Malhorta A. Sleep Disorders Handbook.
10. Fuller PM, Gooley JJ, Saper CB. Neurobiology of the sleep-wake cycle: sleep architecture, circadian regulation, and regulatory feedback. J Biol Rhythms. 2006;21(6):482-493.
11. Harris CD. Neurophysiology of sleep and wakefulness. Respir Care Clin N Am. 2005;11(4):567-586.
12. Macaluso GM, Guerra P, DiGiovanni G, et al. Sleep bruxism is a disorder related to periodic arousals during sleep. J Dent Res. 1998;77(4):565-573.
13. Huynh N, Kato T, Rompré PH, et al. Sleep bruxism is associated to micro-arousals and an increase in cardiac sympathetic activity. J Sleep Res. 2006;15(3):339-346.
14. Ahlberg K, Savolainen A, Könönen M, et al. Bruxism and sleep efficiency measured at home with wireless devices. J Oral Rehabil. May 9, 2008. Epub ahead of print.
15. Smith SS, Dingwall K, Jorgenson G, et al. Associations between the use of common medications and sleep architecture in patients with untreated obstructive sleep apnea. J Clin Sleep Med. 2006;2(2):156-162.
16. Kushida CA, Littner MR, Morgenthaler T, et al. Practice parameters for the indications for polysomnography and related procedures: an update for 2005. Sleep.2005;28(4):499-521.
17. Russo MB. Sleep stage scoring. March 2007. eMedicine from WebMD Web site. Available at: http://www.emedicine.com/neuro/topic443.htm. Accessed May 12, 2008.
18. White DP. The pathogenesis of obstructive sleep apnea: advances in the past 100 years. Am J Respir Cell Mol Biol. 2006;34(1):1-6.
19. Young T, Evans L, Finn L, Palta M. Estimation of the clinically diagnosed proportion of sleep apnea syndrome in middle-aged men and women. Sleep. 1997;20(9):705-706.
20. Collop NA, Anderson WM, Boehlecke B, et al. Clinical guidelines for the use of unattended portable monitors in the diagnosis of obstructive sleep apnea in adult patients. Portable Monitoring Task Force of the
21. Dement WC, Koenigsberg R. Winds of change in sleep medicine. Sleep Rev. 2008;9(5):12-17.
22. Haponik EF, Frye AW, Richards B, et al. Sleep history is neglected diagnostic information. Challenges for primary care physicians. J Gen Intern Med. 1996;11(12):759-761.
23. Bailey DR, Attanasio R. Dentistry's role in the management of sleep disorders. Recognition and management. Dent Clin North Am, 2001;45(4):619-630.
24. Friedlander AH, Friedlander IK, Pogrel MA. Dentistry’s role in the diagnosis and co-management of patients with sleep apnoea/hypopnoea syndrome. Br Dent J. 2000;189(2):76-80.
25. Tsai WH, Remmers JE, Brant R, et al. A decision rule for diagnostic testing in obstructive sleep apnea. Am J Respir Crit Care Med. 2003;167(10):1427-1432.
26. Flemons WW,
27. Lussi A, Hellwig E. Risk assessment and preventive measures. Monogr Oral Sci, 2006;20:190-191.
28. Miyawaki S, Tanimoto Y, Araki Y, et al. Association between nocturnal bruxism and gastroesophageal reflux. Sleep. 2003;26(7):888-892.
29. Ali DA, Brown RS, Rodriquez LO, et al. Dental erosion caused by silent gastrosophageal reflux disease. J Am Dent Assoc. 2002;133(6):734-737.
30. Ahmed M, Patel NP, Rosen I. Portable monitors in the diagnosis of obstructive sleep apnea. Chest. 2007;132(5):1672-1677.
31. Kushida CA, Morgenthaler TI, Littner MR, et al. Practice parameters for the treatment of snoring and obstructive sleep apnea with oral appliances: an update for 2005. Sleep. 2006; 29(2):240-243.
32. Verse T, Pirsiq W, Stuck BA, et al. Recent developments in the treatment of obstructive sleep apnea. Am J Respir Med. 2003;2(2):157-168.
33. Yetkin O, Kunter E, Gunen H. CPAP compliance in patients with obstructive sleep apnea syndrome. Sleep Breath, 2008 May 15. Epub ahead of print.
34. Kribbs NB, Pack AI, Kline LR, et al. Objective measurement of patterns of nasal CPAP use by patients with obstructive sleep apnea. Am Rev Respir Dis. 1993;147(4):887-895.
35. Reeves-Hoche MK, Meck R, Zwillich CW. Nasal CPAP: an objective evaluation of patient compliance. Am J Respir Crit Care Med. 1994;149(1):149-154.
36. Waldhorn RE, Herrick TW, Nguyen MC, et al. Long-term compliance with nasal continuous positive airway pressure therapy of obstructive sleep apnea. Chest. 1990;97(1):33-38.
37. van den Broek E, Richard W, van Tinteren H, de Vries N. UPPP combined with radiofrequency thermotherapy of the tongue base for the treatment of obstructive sleep apnea syndrome. Eur Arch Otorhinolaryngol. May 15,2008. Epub ahead of print.
38. Sundaram S, Bridgman SA, Lim J, et al. Surgery for obstructive sleep apnea. Cochrane Database Syst Rev. 2005(4):CD001004.
39.
40. Schmidt-Nowara W, Lowe A, Weigand L, et al. Oral appliances for the treatment of snoring and obstructive sleep apnea: a review. Sleep. 1995;18(6):501-510.
41. Cohen R. Obstructive sleep apnea: oral appliance therapy and severity of condition. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1998;85(4):388-392.
42. Wade PS. Oral appliance therapy for snoring and sleep apnea: preliminary report on 86 patients fitted with an anterior mandibular positioning device, the Silencer. J Otolaryngol. 2003;32(2):110-113.
43. Guerrero M, Lepler L, Kristo D. The upper airway resistance syndrome masquerading as nocturnal asthma and successfully treated with an oral appliance. Sleep Breath. 2001;5(2):93-96.
44. Ivanhoe JR, Attanasio R. Sleep disorders and oral devices. Dent Clin North Am. 2001; 45(4):733-758.
45. Clark GT, Blumenfeld I, Peled R, et al. A crossover study comparing the efficacy of continuous positive airway pressure with anterior mandibular positioning devices on patients with obstructive sleep apnea. Chest. 1996;109(6):1477-1483.
46.
47.
48. Zozula R, Rosen R. Compliance with continuous positive airway pressure therapy: assessing and improving treatment outcomes. Curr Opin Pulm Med. 2001;7(6):391-398.
49. Ryan CF, Love LL, Peat D, et al. Mandibular advancement oral appliance therapy for obstructive sleep apnoea: effect on awake calibre of the velopharynx. Thorax. 1999;54(11):972-977.
About the Author:
Barry Glassman, DMD
Private Practice Limited to Chronic Pain Management, Temporomandibular Joint Dysfunction, and Dental Sleep Medicine
| Figure 1 The Epworth Sleepiness Test is used to evaluate excessive daytime sleepiness. | Figure 2 K complexes and sleep spindles are EEG patterns specific to Stage 2 sleep. | Figure 3 A polysomnogram records 16 or more channels of a patient’s sleep physiology while in a sleep laboratory. |
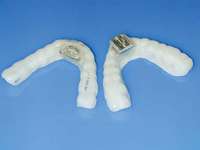
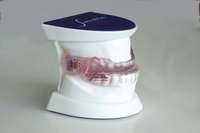
| Figure 4 Sleep-disordered breathing is a continuum. Diagnosis is required before treatment is considered. | Figure 5 Obstructive sleep disorders commonly have obstructions at more than one level. | Figure 6 The SomnoDENT (SomnoMED, Crows Nest, NSW, |
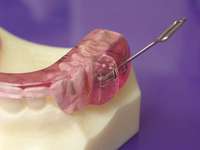
| Figure 7 The author’s preference is to use appliances with anterior midpoint stops for parafunctional control. | Figure 8 The TAP® III (Comfort Acrylics, Inc, Camas,WA) is a Thornton Anterior Positioner. This version has thermacryl on the interior that allows for easy adaptation with dental changes. | Figure 9 The TAP III is titratable by the patient. Titration of this appliance will control the protrusion of the mandible. Both the TAP and SomnoDENT provide patient-friendly titration, which is extremely helpful as adjustments are made to assure comfort and yet provide adequate muscular tension. |
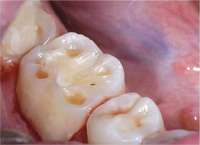
| Figure 10 Dentinal pooling and incisal scooping may be among the first signs of GERD, which can be a precursor to Barrett’s Syndrome. | Figure 11 Ambulatory tests can be used in medicine and dentistry. Dentists use them for screening before referring to a medical professional or making decisions about titration of an oral appliance. | Figure 12 The uvulopalatalphyrgolplasty procedure can be effective for sleep-disordered breathing but the lack of predictability of the success of the surgery, combined with a difficult postsurgical course, has decreased the frequency the surgery is recommended. |
|
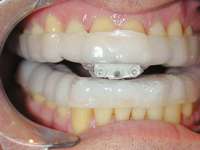
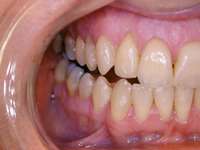
Figure 13 The SomnoDENT is made by SomnoMED. The quality control of this appliance is excellent, and it is durable and comfortable. | Figure 14 One of the complications of oral appliance therapy can be the development of posterior open bite. It is not common, but when it occurs usually function is not altered. It is essential that an informed risk/benefit decision is made. | Figure 15 A key is used by the patient who is given a specific schedule to titrate the appliance in the anterior direction. |
Table: Screening Questions
- Do you snore?
- Do you have high blood pressure?
- Has anyone reported that you choke or gasp for air while sleeping?
- What is your neck size? (inches)
- Do you wake refreshed?
- Are you excessively tired during the day?