You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
ABSTRACT
Never before in dentistry has a group of drugs rocketed into the limelight. The sudden rise to infamy of the group of drugs called bisphosphonates is unfortunately because of the serious complication of osteonecrosis of the jaws. This article has three simple goals. The first two are to give readers an understanding of bisphosphonates and describe their common uses by providing a chemical description of the drugs, how the drugs work, why the drugs are prescribed, and why the drugs cause complications. The third goal is to give readers suggestions on managing their patients to prevent or minimize oral complications.
Bisphosphonates are a group of drugs with a phosphorus-carbon-phosphorus (P-C-P) backbone.1 This backbone makes the drugs unable to be hydrolyzed and is responsible for their long activity. The carbon in the backbone is bonded to a hydroxyl (-OH) group on one side which, in turn, is responsible for binding to hydroxyapatite, a major component of bone. The carbon in the backbone also is bonded to two side chains. The side chain containing nitrogen is the most potent of the bisphosphonates and is associated with the most complications. This simple chemical makeup of these drugs gives them a half-life of more than 10 years.
Bisphosphonates are concentrated in bone and, after the osteoclasts ingest the drugs, kill or disable the osteoclasts.2 The drugs can interfere with the energy production of osteoclasts or with their biosynthetic pathways. Osteoclasts are boneremodeling cells that remove old or damaged bone. They are essential in bone physiology.
When patients begin using bisphosphonates, the drug is concentrated in the oral cavity because the oral cavity has a very high turnover rate from the breakdown and redeposition of bone.3 Animal studies have put this rate as high as 35% of the bone per year. The high concentration of the drug kills so many osteoclasts that the bone cannot renew itself properly. The bone then becomes necrotic and, when exposed to trauma, is unable to heal.
USES OF BISPHOSPHONATES
There are many diseases in which osteoclasts cause the body harm and for which bisphosphonates are prescribed. Table 1 lists all current bisphosphonates and their primary uses.
Osteoporosis, one of the most common of these diseases, is a disease of bone metabolism. As people age, osteoblasts, which are the bone-making cells, cannot keep up with osteoclasts, the bone-taking cells. This imbalance causes the bone to lose its density and become brittle.4 Less dense bone is very susceptible to fractures, which often occur in the hip or the spinal column. There are about 1.5 million fractures a year in the United States and a large percentage of these fractures result in death. Bisphosphonates are very useful in the management of osteoporosis.5 They reverse the decrease in bone mineral density and reduce the frequency of fractures by slowing the imbalance between osteoclasts and osteoblasts.5
Another disease in which osteoclasts cause the body harm is cancer. When someone has metastatic cancer, the cancer cells secrete the substance receptor activator of nuclear factor kappa B ligand (RANKL).6 RANKL is an osteoclast stimulator. This stimulator causes osteoclasts to break down bone, thus allowing cancer cells to grow into the spaces created, ultimately causing a fracture. Metastatic cancer cells also secrete a substance similar to parathyroid hormone, which further causes the osteoclasts to break down bone.
Because bone breakdown is extensive, patients with metastatic cancer often become hypercalcemic. The subsequent increase of calcium often causes muscle pain, confusion and, in some cases, cardiac arrest. Because metastatic cancer poses these risks, if physicians even suspect metastases, they prescribe bisphosphonates. Paget’s disease is another disease in which osteoclasts cause the body harm. Although the cause of this disease is not fully understood, a virus is suspected. With this disease, the body undergoes a tremendous amount of bone remodeling. This remodeling is so severe and so rapid that the body cannot do it properly. The body, thus, makes soft and immature bone, known as cotton-wool bone. Bisphosphonates have been very successful in managing this disease. Paget’s disease was one of the first diseases for which bisphosphonates were used.
Osteoclasts also cause the body harm in patients with multiple myeloma. This disease is a cancer of plasma cells. With this disease, plasma cells secrete interleukin-6, which stimulates osteoclasts.7 This stimulation of osteoclasts causes bone breakdown and the resulting classic “punchedout” lesions often are seen on radiographs of the bone.
Many off-label uses of bisphosphonates exist, including treatment of juvenile osteoporosis, Gaucher’s disease, fibrous dysplasia, and osteogenesis imperfecta. Because dentists may treat many patients using these drugs, it is important that they understand how to manage these patients to prevent, or at least minimize, oral complications.
COMPLICATIONS
Bisphosphonate-related osteonecrosis of the jaws (BRONJ) is characterized by the presence of exposed bone in the oral cavity for more than 8 weeks in a patient who has been taking bisphosphonates and who has not had any radiation therapy in the oral cavity3 (Figure 1). The risk of BRONJ from the use of oral bisphosphonates is very low, currently about 1%.8 Most researchers believe this incidence is so low because it takes many years for the drug to saturate the bone. Most cases of BRONJ from oral bisphosphonates occur after at least 3 years of drug use.9 However, a recent study by Cartsos et al of over 700,000 medical claims found no increased incidence of inflammatory conditions, including osteonecrosis.10
When given in the intravenous (IV) form, it can take as little as 1 year for the drug to saturate bone, yielding a much higher incidence of BRONJ.11 Many researchers put the risk of IV BRONJ at 5% to 15%.3
PATIENT MANAGEMENT
The first step in understanding how to manage dental patients who use bisphosphonates is to read current literature. Because information about these drugs is constantly evolving, keeping up to date is essential. However, it is important not to ignore or overreact to the risks bisphosphonates pose. In 2006, the American Dental Association (ADA) Council on Scientific Affairs8 published their expert panel’s recommendations on how to manage patients on oral bisphosphonates. The article also provides an Internet address to keep up with changes.Another noteworthy article is the American Academy of Oral Medicine’s position paper on managing patients on bisphosphonates by Migliorati et al.14 The article gives clear information on how to manage patients on oral and IV bisphosphonates.
Another resource for dentists is Oral & Intravenous Bisphosphonate-Induced Osteonecrosis of the Jaws: History, Etiology, Prevention, and Treatment by Robert Marx.3 In 2003, Marx was first to describe BRONJ,12 and his book gives readers short and concise information. The second step in managing patients is to take a proper medical history. The medical history form should include questions about bisphosphonate use. Many patients do not think it is important to disclose all of the drugs they take. If a patient checks “no” in the medical history for bisphosphonate use, then due diligence has been performed. If a patient checks “yes,” then move to the next step in patient management.
The third step in management is a consent form,13 such as the one provided on the ADA’s Web site (www.ada.org/prof/resources/topics/osteonecrosis.asp). Consent forms are the law in many states. Using a consent form for patients who are taking bisphosphonates gives the dentist and patient an opportunity to talk about the drug and its possible side effects. BRONJ is the main side effect that should be discussed with dental patients. It is important to emphasize to patients that the purpose of the discussion and the consent form is to inform them, not to unnecessarily alarm them. Most patients are appreciative.
IV Bisphosphonate Users
For patients on IV bisphosphonates, after bone is exposed, it is almost impossible to cover.15 Most cases worsen if surgical intervention is performed. Most patients must simply deal with the exposed bone and keep it clean so that bacterial infection does not occur. Figure 2 and Figure 3 show a postmetastatic prostate cancer patient with exposed bone. The treatment plan for this patient was to make a soft-lined denture that would not put pressure on the exposed bone and to check for bacterial infection on a routine basis. When dealing with patients on or about to begin IV bisphosphonate therapy, dentists need to be very astute. As stated earlier, this form of the drug has a much higher incidence of complications. These patients are often very sick and immuno-compromised and, therefore, must be managed properly. Pre-IV bisphosphonate patients (ie, those not yet receiving the drug) are much easier to manage than patients already receiving the drug. First, ask the treating physician, usually an oncologist, if he or she can wait 2 months before the drug is given. If the cancer is severe, it may not be possible to wait. Dentists should make use of whatever time can be made available before bisphosphonate treatment begins.16 This time should be used to get a full-mouth series of radiographs and to remove all dental infections, including all decay. If decay cannot be removed completely, the tooth should be considered for extraction. Before IV therapy begins, patients are at a lower risk for extraction complications than they are during bisphosphonate therapy.
If a patient requires root canal therapy, it must be performed completely and properly. The tooth must be instrumented properly and no apical extension should occur. Any overextension can cause trauma and increase the risk of BRONJ. If a patient has periodontal diseases, they must be thoroughly treated. If any signs of infection (bleeding, swelling, and/or abnormal color) are present after scaling and root planing, further treatment is indicated. The patient likely requires periodontal surgery. If a patient wears removable prosthetic appliances, they must be checked and any sharp edges or areas that rub must be eliminated.17 If problems with any appliance cannot be eliminated, the patient should be advised not to wear that appliance until IV therapy is finished. After IV therapy is complete, new appliances can be fabricated. Multilobulated tori also can trigger BRONJ and should be removed if time permits.18 These tori are triggers because they have high bone turnover rates, thin overlying tissue, and are traumatized easily.
After all dental infections have been removed, patients can initiate IV therapy. Dentists should follow-up regularly with these patients during their bisphosphonate therapy, preferably once a month. After patients are off bisphosphonate therapy for 2 months, their radiograph series should be updated. The radiographs should be viewed for signs of sclerosis of the lamina dura or widening of the periodontal ligament because these are usually the first places where signs of bisphosphonate toxicity and osteonecrosis can be seen.19
Intra-IV therapy patients (ie, those already receiving IV bisphosphonate therapy) are difficult to treat and should be treated cautiously. The most important thing with intra-IV patients is to treat them as atraumatically as possible and avoid surgery at all costs.20 For example, a root canal can be performed as an alternative to an extraction. Treat periodontal patients who do not respond to nonsurgical treatment with chlorhexidine rinse and antibiotics.After patients are no longer receiving the drug, still proceed with care and avoid surgery. If surgery is unavoidable, consult with the treating physician and make sure the patient understands all of the risks associated with surgery.
Oral Bisphosphonate Users
Dentists are more likely to encounter patients taking oral bisphosphonates. Most often, these patients are identified from their medical histories. After patients have been identified as oral bisphosphonate users, they should be given a consent form. Asking them how long they have been on the drug is important because the longer they have been taking the drug, the greater their risk for BRONJ. If a patient has been taking the drug for less than 3 years, then that patient, most likely, can be treated normally. However, with these patients, they should be informed of the small risk of BRONJ for every person taking bisphosphonates and that the risk increases after 3 years of drug use. This group of patients is also more likely to be taking steroids, which have been found to be a risk factor for osteonecrosis.
All decay and infections in the oral bisphosphonate user should be removed, explaining to the patient that such infections can increase the likelihood of BRONJ. Most researchers find periodontal diseases and extractions as the most frequent comorbidity linked to BRONJ, although spontaneous osteonecrosis is also possible.21
Most operative procedures can be done in patients taking oral bisphosphonates, but with special care to limit the amount of trauma to the gingiva and bone. If a patient needs an extraction and he or she has been on the drug for more than 3 years, it may be preferable to perform a root canal, cut the crown off the tooth, and leave the root tip. If a patient on oral bisphosphonates needs surgery, the sextant approach is recommended.
Under the sextant approach, one quadrant is treated and the patient is left to heal for 2 months before proceeding to any additional quadrants. While the patient heals, a 10-day course of antibiotics and 2 months of twice-daily chlorhexidine rinsing may be prescribed. Figure 4 shows a patient who was taking oral bisphosphonates for 3 years and was treated with the sextant approach. As seen in the radiograph, the patient has one molar in the lower right quadrant. This tooth was extracted and the patient was given 2 months to heal (Figure 5 and Figure 6).The patient was placed on antibiotics and chlorhexidine rinse. After 2 months, no signs of BRONJ were observed, and the anterior teeth were scheduled for extraction. They were extracted as atraumatically as possible, and the site was sutured (Figure 7 and Figure 8).The patient then was given another 2 months to heal, following the previous protocol.After 2 months of healing, no BRONJ was observed (Figure 9).
Then, patient was able to move forward with the prosthesis. Figure 10 shows a periodontal patient who was taking oral bisphosphonates for osteoporosis. The patient had full-mouth scaling and root planing, then was given 2 months to heal and instructed to rinse with chlorhexidine twice daily. After no signs of BRONJ were observed, the patient had full-mouth osseous surgery on her few remaining teeth in the mandible. The surgery was performed all at once and not according to the sextant approach because the patient was on a heart and lung transplant list and was moved up the list while healing from the scaling and root planing.
The patient also had extensive bone loss in her anterior mandible (Figure 11).To avoid performing extractions, a simple composite splint was placed between the partial dentures (Figure 12 and Figure 13). The splint reduced the mobility of the teeth and made them less likely to be knocked out during any surgical intubation procedure. As stated earlier, spontaneous osteonecrosis is also possible. Figure 14 shows a had noticed the foreign material, but chose not to act on it. When the material was removed in the dental office (Figure 15 and Figure 16), blood streamed from the nose, so severely that the patient had to be sent to an ear, nose, and throat (ENT) physician. The ENT took a computed tomography scan, which showed a large radiolucency in the upper left quadrant where the foreign material once had been (Figure 17). A review of the radiograph (Figure 18) showed that bone in the maxillary arch had decreased mineral density compared with the mandible. The biopsy report found necrotic bone with organisms consistent with actinomyces. These findings were consistent with biopsies found in the literature.22 When the patient returned for a 1-week follow-up, she still had signs of infection and the site had a foul odor and a discharge (Figure 19). The patient was informed that she should follow up with the ENT and return to the dental office after the infection had been resolved.
CONCLUSION
This article presented three simple steps to help dentists manage patients who use bisphosphonates. The first step is stay current on the relevant literature given the constant changes in researchers’ understanding of bisphosphonates. The second step is to have a current, signed medical history for each patient, with updates as needed. This is necessary because most patients taking bisphosphonates are identified from their medical history. The third step is to use a consent form for all patients taking bisphosphonates. The form is recommended for two main reasons: (1) it gives the patient and dentist the opportunity to talk about bisphosphonates and their risk factors; and (2) it gives the dentist important legal coverage if some adverse event occurs. Any patient with signs of previously undiagnosed BRONJ should be referred to an oral surgeon. It should be noted that although these steps may prove useful to practitioners, they are solely the author’s opinion and are not intended as scientific conclusions based on treatment outcome studies.
Many in the dental community are working to create a standard of care for treating bisphosphonate users. The most promising is the CTx serum test, but no dental association has yet endorsed this test. Given that there is no established standard of care, each practitioner will need to decide what approach to take, including whether to follow the CTx serum test or some other protocol. Ultimately, every patient must know the potential side effects from taking bisphosphonates and accept the resulting risk associated with any dental treatment.
REFERENCES
1. Graham JW. Bisphosphonates and orthodontics: clinical implications. J Clin Orthod. 2006;40(7):424-428.
2. Wynn RL. Oral bisphosphonates and osteonecrosis of the jaw. Gen Dent. 2007;55(1):8-10.
3. Marx RE. Oral & Intravenous Bisphosphonate-Induced Osteonecrosis of the Jaws: History, Etiology, Prevention, and Treatment. Hanover Park, IL Quintessence 2007;1-150.
4. Leite AF, Figueiredo PT, Melo NS, et al. Bisphosphonate-associated osteonecrosis of the jaws, report of a case and literature review. Oral Sur Oral Med Pathol Oral Radio lEndod. 2006;102(1):14-21.
5. Grbic JT, Landesberg R, Lin SQ, et al. Incidence of osteonecrosis of the jaw in women with postmenopausal osteoporosis in the Health Outcomes and Reduced Incidence With Zoledronic Acid Once Yearly Pivotal Fracture Trial. J Am Dent Assoc. 2008;139(1):32-40.
6. Ruggiero SL, Drew SJ. Osteonecrosis of the jaws and bisphosphonate therapy [published erratum in: J Dent Res. 2007;86(12):1223]. J Dent Res. 2007;86(11): 1013-1021.
7. Zak M, Spina AM, Spinasse RP, et al. Bisphosphonates and the dental patient: part 1. Compend Contin Educ Dent. 2007; 28(8):464-469.
8. American Dental Association Council on Scientific Affairs. Dental management of patients receiving oral bisphosphonate therapy: expert panel recommendations. J Am Dent Assoc. 2006;137(8):1144-1150.
9. Marx RE, Cillo JE Jr, Ulloa JJ. Oral bisphosphonate-induced osteonecrosis: risk factors, prediction of risk using serum CTX testing, prevention, and treatment. J Oral Maxillofac Surg. 2007;65(12):2397-2410.
10. Cartsos VM, Zhu S, Zavras AI. Bisphosphonate use and the risk of adverse jaw outcomes: a medical claims study of 714,217 people. J Am Dent Assoc. 2008;139(1):12-15.
11. Koka S, Clarke BL, Amin S, et al. Oral bisphosphonate therapy and osteonecrosis of the jaw: what to tell the concerned patient. Int J Prosthodont. 2007;20(2):115-122.
12. Thoman RD. Osteonecrosis: what dentists need to know. Hawaii Dent J. 2006;37(6):9-13.
13. Bittinger A. Consider informed consent policy for treating bisphosphonate patients. Today’s FDA. 2007;19(1):9-10.
14. Migliorati CA, Casiglia J, Epstein J, et al. Managing the care of patients with bisphosphonate-associated osteonecrosis: an American Academy of Oral Medicine position paper [published erratum in: J Am Dent Assoc. 2006;137(1):26]. J Am Dent Assoc. 2005;136(12):1658-1668.
15. Greenberg MS. Intravenous bisphosphonates and osteonecrosis [editorial]. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2004;98(3):259-260.
16. Ficarra G, Beninati F, Rubino I, et al. Osteonecrosis of the jaws in periodontal patients with a history of bisphosphonates treatment. J Clin Periodontol. 2005;32(11): 1123-1128.
17. Bagán J, Blade J, Cozar JM, et al. Recommendations for the prevention, diagnosis, and treatment of osteonecrosis of the jaw (ONJ) in cancer patients treated with bisphosphonates. Med Oral Patol Oral Cir Buccal. 2007;12(4):E336-340.
18. Zak M, Spina AM, Spinasse RP, et al. Bisphosphonates and the dental patient: part 2. Compend Contin Educ Dent. 2007;28(9):510-516.
19. Ruggiera Sl, Fantasia J, Carlson E. Bisphosphonate-related osteonecrosis of the jaw: background and guidelines for diagnosis and management. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006; 102(4):433-441.
20. Braun E, Iacono VJ. Bisphosphonates: case reports of nonsurgical periodontal therapy and osteochemonecrosis. Int J Periodontics Restorative Dent. 2006;26(4):315-319.
21. Levin L, Laviv A, Schwartz-Arad D. Denture-related osteonecrosis of the maxilla associated with oral bisphosphonate treatment. J Am Dent Assoc. 2007;138(9)1218-1220.
22. Hellstein JW, Marek CL. Bisphosphonate osteochemonecrosis (bis-phossy jaw): is this phossy jaw of the 21st century? J Oral Maxillofac Surg. 2005;63(5):682-689.
|
|
| 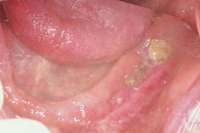 Figure 3 Figure 3 |
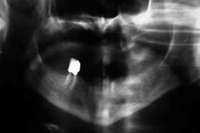 Figure 4 Radiograph of oral bisphosphonate user. Figure 4 Radiograph of oral bisphosphonate user. | 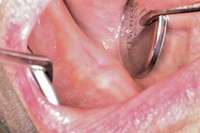 Figure 5 and Figure 6 After 2 months of healing, the lower right quadrant showed no signs of BRONJ and the anterior teeth were scheduled for extraction. Figure 5 and Figure 6 After 2 months of healing, the lower right quadrant showed no signs of BRONJ and the anterior teeth were scheduled for extraction. | 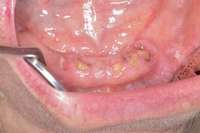 Figure 6 Figure 6 |
|
|
| 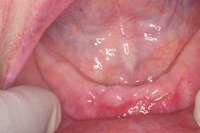 Figure 9 For this quadrant, no signs of BRONJ were observed after 2 months of healing. Figure 9 For this quadrant, no signs of BRONJ were observed after 2 months of healing. |
|
|
| 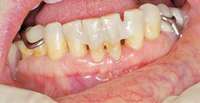 Figure 12 Facial view of composite splint. Figure 12 Facial view of composite splint. |
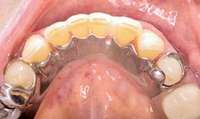 Figure 13 Occlusal view of composite splint. Figure 13 Occlusal view of composite splint. | 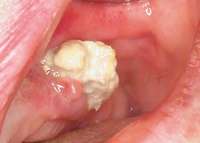 Figure 14 Spontaneous BRONJ. Figure 14 Spontaneous BRONJ. |
|
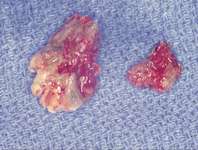 Figure 16 Necrotic bone. Figure 16 Necrotic bone. | 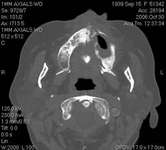 Figure 17 CT scan showing extensive bone loss. Figure 17 CT scan showing extensive bone loss. | 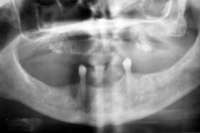 Figure 18 Panoramic radiograph showing decreased bone density. Figure 18 Panoramic radiograph showing decreased bone density. |
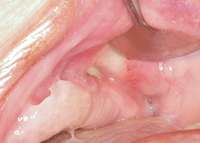 Figure 19 Infection seen after the biopsy. Figure 19 Infection seen after the biopsy. |
About the Author
Wilkie J. Stadeker, DDS
Private Practice limited to Periodontics, Marietta, Georgia
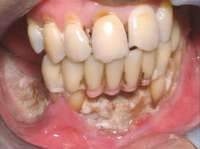 Figure 1 Exposed bone in the mandibular jaw that is characteristic of BRONJ. Photo courtesy of Robert Marx, DDS.
Figure 1 Exposed bone in the mandibular jaw that is characteristic of BRONJ. Photo courtesy of Robert Marx, DDS.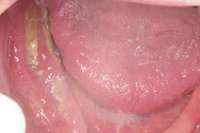 Figure 2 and Figure 3 Postmetastatic cancer patient with BRONJ (A) in the lower right quadrant and (B) in the lower left quadrant.
Figure 2 and Figure 3 Postmetastatic cancer patient with BRONJ (A) in the lower right quadrant and (B) in the lower left quadrant.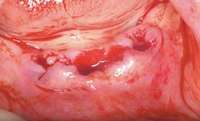 Figure 7 Anterior extraction sites after atraumatic removal.
Figure 7 Anterior extraction sites after atraumatic removal.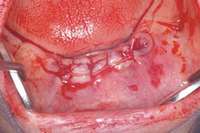 Figure 8 Sutures were placed after extraction.
Figure 8 Sutures were placed after extraction.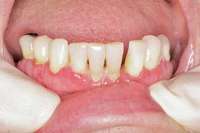 Figure 10 Patient who takes oral bisphosphonates and suffers from periodontal disease.
Figure 10 Patient who takes oral bisphosphonates and suffers from periodontal disease.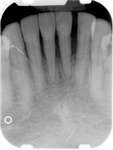 Figure 11 The radiograph shows 80% bone loss.
Figure 11 The radiograph shows 80% bone loss.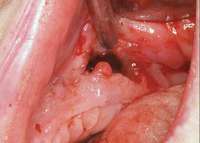 Figure 15 Removal of the necrotic bone material.
Figure 15 Removal of the necrotic bone material.