You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
High rates of opioid pre-scribing have been linked to the growing epidemic of opioid abuse in the United States, which is a significant public health concern.1-4 Unfortunately, oral healthcare practitioners are more likely to prescribe opioids than primary care physicians, and areas with a higher number of dentists per capita are associated with increased opioid prescribing rates.5,6 In fact, dentists are responsible for 6.4% of all outpatient immediate-release opioid prescriptions in the United States.6,7 An article published more than 7 years ago in the Journal of the American Dental Association identified, even at that time, that "the appropriate use of opioids requires dentists to follow responsible and tailored prescribing practices to provide adequate pain control while limiting opportunities for abuse and diversion."8 Since then, many publications have come forward with additional statistics to highlight this issue, but very few of these have been directional in helping the frontline prescribing oral healthcare practitioners choose better alternatives to manage their patients' pain.9-15
Postoperative Analgesia
In a 2010 publication, "Appropriate analgesic prescribing for the general dentist," the mnemonic "2-4-24" was introduced to help practitioners remember the postoperative prescription of choice for acute nociceptive orofacial pain.16 This memory aid refers to, "2 drugs, in 4 doses, for 24 hours," which indicates a combination of ibuprofen 600 mg and acetaminophen 650 mg to be administered every 6 hours for 24 hours. This dosing mnemonic is especially useful because it is both effective and safe. For the treatment of acute pain, the maximum recommended dose that should not be exceeded during a 24-hour period is 2,400 mg for ibuprofen and 3,000 mg for acetaminophen. The combination of a nonsteroidal anti-inflammatory drug (NSAID), such as ibuprofen, and acetaminophen administered every 6 hours for at least 24 hours has been recognized and validated by both individual studies and meta-analyses as the postoperative prescription of choice for acute nociceptive orofacial pain.16-19 An understanding of the inflammatory process, the pharmacokinetics of these medications, and routine dosing (as opposed to prn or "as needed" dosing) during the initial 24-hour postoperative period is required to ensure success. In a majority of cases, if the patient is compliant with these four doses of two different medications, he or she may not require any further analgesic medication beyond the first 24 hours. These doses can be administered every 6 hours, either together or in a staggered fashion, based on dentist and patient preference (eg, 650 mg of acetaminophen now, then 600 mg of ibuprofen 3 hours later, then 650 mg of acetaminophen 3 hours after that, and so on for the first 24-hour postoperative period). The staggered approach is sometimes valuable for patients known as "clock watchers" for whom more frequent medication administration may be more psychologically beneficial following dental surgery. These two medications work by different mechanisms of action, and compliance is the key to the success of this analgesic regimen, which may require that patients set alarms so that they do not miss the prescribed doses during the initial 24-hour postoperative period. After the initial 24-hour postoperative period, patients can reduce their regimen to a prn administration of either drug by itself or in combination. However, if patients still require routine pain medication after the initial 48-hour period following the dental procedure despite excellent compliance, reexamination by the dental practitioner should be considered to rule out confounders such as wound disturbances or infection.
Although dexamethasone was approved by the US Food & Drug Administration in 1958 under the brand name Decadron®, the first published reference discussing glucocorticoid use in dentistry utilized other corticosteroids, such as cortisone and prednisolone.20,21 The first recorded use of dexamethasone for endodontic procedures was not until 1968, and its use continues to be an important pain control modality today.22 Dexamethasone comes in both oral and injectable formulations. The oral formulation is especially useful for postoperative analgesia and is more commonly recognized as a 6-, 10-, or 13-day tapered regimen (eg, Dexpak®).23 These tapered regimens are not required to prevent adrenal insufficiency because it typically takes 14 days of supraphysiological doses of exogenous glucocorticoids to suppress the hypopituitary axis. As was demonstrated in a recent systematic review, a simpler strategy would be to consider prescribing 4 to 8 mg of oral dexamethasone to be taken twice a day for up to 48 hours after dental surgery.24 Alternatively, dexamethasone has also been successfully used as a postoperative intramuscular injection for the control of pain, swelling, and trismus after third molar surgery, which was demonstrated in a systematic review and meta-analysis by Fernandes and colleagues.25
Preemptive Analgesia
Medications such as acetaminophen and ibuprofen can also be given preoperatively to mitigate postoperative pain and analgesic medication requirements.26,27 A cyclooxygenase-2 (COX-2) selective NSAID, such as celecoxib, is ideally suited for this strategy. When compared with traditional nonselective NSAIDs, celecoxib can preemptively ameliorate the inflammatory response without causing increased bleeding or delayed wound healing postoperatively.28-30 A 400 mg dose of celecoxib administered orally 30 minutes prior to the dental procedure can maximize this potential benefit. For those patients who are currently on an anticoagulant (eg, warfarin, dabigatran, rivaroxaban, apixaban, betrixaban, edoxaban), a COX-2 inhibitor such as celecoxib could further be used to replace the previously described postoperative ibuprofen prescription (ie, 200 mg given every 12 hours for the initial 24-hour postoperative period).
Preemptive analgesia with 4 to 8 mg of a glucocorticoid, such as dexamethasone, taken orally 30 minutes prior to the procedure has also been shown to be an excellent strategy, either alone or in addition to celecoxib.31,32
Intraoperative Analgesia
Intraoperative analgesia is at least a 70-year-old concept in dentistry; lidocaine entered its seventh decade as a part of the United States Pharmacopeia in 2018.33 The opioid epidemic has renewed interest in intraoperative analgesics because they not only help with immediate pain management but also decrease a patient's typical requirement for postoperative narcotics.
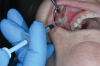
A single intraoperative injection of 4 to 8 mg of dexamethasone administered submucosally adjacent to the area of treatment is an excellent addition to the pain control strategies discussed thus far.16,34 Dan and colleagues had already shown in an earlier meta-analysis that an intraoperative injection of methylprednisolone significantly reduced edema and pain following oral surgery without an increased risk of infection or other adverse effects35; then, in a more recent meta-analysis, Chen and colleagues found that a single intraoperative injection of up to 10 mg of dexamethasone reduced not only early and late edema but also early trismus and pain following third molar extractions.34 Particularly helpful for practitioners is that a single intraoperative injection of dexamethasone can provide analgesia for up to 6 days when compared with oral administration, which provides an approximate 2-day duration of action.36 Many endodontists have been using this strategy successfully for years, but the new evidence helps to support the more routine use of a single intraoperative injection of dexamethasone following other types of dental surgeries. This submucosal injection, which is similar to a typical infiltration approach, is easily administered and should be delivered into a location of the mouth that has already been anesthetized to improve the patient's comfort (Figure 1).
Because patient safety is always the foremost concern, an additional benefit of this single intraoperative injection of dexamethasone is that it is accompanied by very few contraindications. Although it is not recommended for patients who are pregnant or lactating or who have uncontrolled diabetes, the chance of experiencing a life-threatening reaction to a single intraoperative dose of dexamethasone is highly unlikely. In their systematic review, Chen and colleagues found that none of the included studies addressed the safety of dexamethasone; however, none of the studies reported any severe complications either.34
Conclusion
Recent data reveal that oral healthcare practitioners contribute to the overprescription and overutilization of immediate-release opioid analgesics to manage pain. Although there is a dearth of reports and data, few research articles make suggestions for effective alternative strategies to include drugs, doses, and durations. The combination of 600 mg of an NSAID (eg, ibuprofen) and 650 mg of acetaminophen administered every 6 hours for at least 24 hours has been recognized and validated by individual studies and meta-analyses alike as the postoperative prescription of choice for acute nociceptive orofacial pain. The addition of 4 to 8 mg of oral dexamethasone for preemptive analgesia or 4 to 8 mg of oral dexamethasone twice daily for up to 48 hours after dental surgery also have demonstrated high levels of effectiveness in a recent systematic review on this subject. Finally, a single intraoperative submucosal injection of dexamethasone has been shown to further improve the pain, edema, and trismus that are experienced after dental procedures. Although each of these strategies, either alone or in combination, has greatly aided the clinical management of orofacial pain, all offer the additional benefit of being non-opioid solutions that can be administered to some of the most difficult to manage patients.
Authors' note: The views expressed in this article are those of the authors and do not necessarily reflect those of Premier Dental Products Company, the Creighton University School of Dentistry, the Rutgers School of Dental Medicine, or Vizient, Inc.
About the Authors
Mark Donaldson, PharmD
Fellow
American Society of Health-System Pharmacists
Fellow
American College of Healthcare Executives
Associate Principal
Vizient Pharmacy Advisory Solutions
Irving, Texas
Clinical Professor
University of Montana
School of Pharmacy
Missoula, Montana
Jason H. Goodchild, DMD
Vice President, Clinical Affairs
Premier Dental Products Company
Plymouth Meeting, Pennsylvania
Associate Clinical Professor
Department of Oral and Maxillofacial Surgery
Creighton University
School of Dentistry
Omaha, Nebraska
Adjunct Assistant Professor
Division of Oral Diagnosis
Department of Diagnostic Sciences
Rutgers University
School of Dental Medicine
Newark, New Jersey
References
1. Kolodny A, Courtwright DT, Hwang CS, et al. The prescription opioid and heroin crisis: a public health approach to an epidemic of addiction. Annu Rev Public Health. 2015;36:559-574.
2. U.S. Department of Health & Human Services. HHS acting secretary declares public health emergency to address national opioid crisis. U.S. Department of Health & Human Services website. https://www.hhs.gov/about/news/2017/10/26/hhs-acting-secretary-declares-public-health-emergency-address-national-opioid-crisis.html. Published October 26, 2017. Accessed April 12, 2019.
3. U.S. Department of Health & Human Services. What is the U.S. opioid epidemic? U.S. Department of Health & Human Services website. https://www.hhs.gov/opioids/about-the-epidemic/index.html. Reviewed January 22, 2019. Accessed April 15, 2019.
4. National Institute on Drug Abuse. America's addiction to opioids: heroin and prescription drug abuse. National Institute on Drug Abuse website. https://www.drugabuse.gov/about-nida/legislative-activities/testimony-to-congress/2014/americas-addiction-to-opioids-heroin-prescription-drug-abuse. Published May 14, 2014. Accessed April 15, 2019.
5. Guy GP Jr, Zhang K, Bohm MK, et al. Vital signs: changes in opioid prescribing in the United States, 2006-2015. MMWR Morb Mortal Wkly Rep. 2017;66(26):697-704.
6. Ringwalt C, Gugelmann H, Garrettson M, et al. Differential prescribing of opioid analgesics according to physician specialty for Medicaid patients with chronic noncancer pain diagnoses. Pain Res Manag. 2014;19(4):179-185.
7. Levy B, Paulozzi L, Mack KA, et al. Trends in opioid analgesic-prescribing rates by specialty, U.S., 2007-2012. Am J Prev Med. 2015;49(3):409-413.
8. Denisco RC, Kenna GA, O'Neil MG, et al. Prevention of prescription opioid abuse: the role of the dentist. J Am Dent Assoc. 2011;142(7):800-810.
9. Moore PA, Ziegler KM, Lipman RD, et al. Benefits and harms associated with analgesic medications used in the management of acute dental pain: an overview of systematic reviews. J Am Dent Assoc. 2018;149(4):256-268.
10. Moore PA, Dionne RA, Cooper SA, et al. Why do we prescribe Vicodin? J Am Dent Assoc. 2016;147(7):530-533.
11. McCauley JL, Hyer JM, Ramakrishnan VR, et al. Dental opioid prescribing and multiple opioid prescriptions among dental patients: administrative data from the South Carolina prescription drug monitoring program. J Am Dent Assoc. 2016;147(7):537-544.
12. Lutfiyya MN, Gross AJ, Schvaneveldt N, et al. A scoping review exploring the opioid prescribing practices of US dental professionals. J Am Dent Assoc. 2018;149(12):1011-1023.
13. Gupta N, Vujicic M, Blatz A. Opioid prescribing practices from 2010 through 2015 among dentists in the United States: what do claims data tell us? J Am Dent Assoc. 2018;149(4):237-245.
14. Gupta N, Vujicic M, Blatz A. Multiple opioid prescriptions among privately insured dental patients in the United States: evidence from claims data. J Am Dent Assoc. 2018;149(7):619-627.
15. Koppen L, Suda KJ, Rowan S, et al. Dentists' prescribing of antibiotics and opioids to Medicare Part D beneficiaries: medications of high impact to public health. J Am Dent Assoc. 2018;149(8):721-730.
16. Donaldson M, Goodchild JH. Appropriate analgesic prescribing for the general dentist. Gen Dent. 2010;58(4):291-297.
17. Becker DE. Pain management: part 1: managing acute and postoperative dental pain. Anesth Prog. 2010;57(2):67-78; quiz 79-80.
18. Christensen GJ. Pain meds: what works? Clinicians Report. 2015;8(2).
19. Aminoshariae A, Kulild JC, Donaldson M. Short-term use of nonsteroidal anti-inflammatory drugs and adverse effects: an updated systematic review. J Am Dent Assoc. 2016;147(2):98-110.
20. Schroeder A. Combination of antibiotics and cortisone in the treatment of root canals. Rev Belge Med Dent. 1965;20(3):291-298.
21. Freedman SD. Combination of prednisolone and vitamin therapy in the control of postoperative edema. J Oral Surg Anesth Hosp Dent Serv. 1960;18:332-335.
22. Estefan-Estefan A. The cortico-steroids in endodontics. ADM. 1968;25(2):141-151.
23. Dexpak [package insert]. Bridgewater, NJ: Valeant Pharmaceuticals Inc; 2006.
24. Varvara G, Bernardi S, Cutilli T, et al. Anti-inflam-matory steroid use in impacted third molar surgery: a systematic review. J Biol Regul Homeost Agents. 2017;31(4):1095-1099.
25. Fernandes IA, de Souza GM, Pinheiro MLP, et al. Intramuscular injection of dexamethasone for the control of pain, swelling, and trismus after third molar surgery: a systematic review and meta-analysis. Int J Oral Maxillofac Surg. 2019;48(5):659-668.
26. Nagendrababu V, Pulikkotil SJ, Veettil SK, et al. Effect of nonsteroidal anti-inflammatory drug as an oral premedication on the anesthetic success of inferior alveolar nerve block in treatment of irreversible pulpitis: a systematic review with meta-analysis and trial sequential analysis. J Endod. 2018;44(6):914-922.e2.
27. Shirvani A, Shamszadeh S, Eghbal MJ, et al. Effect of preoperative oral analgesics on pulpal anesthesia in patients with irreversible pulpitis-a systematic review and meta-analysis. Clin Oral Investig. 2017;21(1):43-52.
28. Boonriong T, Tangtrakulwanich B, Glabglay P, Nimmaanrat S. Comparing etoricoxib and celecoxib for preemptive analgesia for acute postoperative pain in patients undergoing arthroscopic anterior cruciate ligament reconstruction: A randomized controlled trial. BMC Musculoskelet Disord. 2010;11:246. doi: 10.1186/1471-2474-11-246.
29. Al-Sukhun J, Al-Sukhun S, Penttilä H, et al. Preemptive analgesic effect of low doses of celecoxib is superior to low doses of traditional nonsteroidal anti-inflammatory drugs. J Craniofac Surg. 2012;23(2):526-529.
30. Munteanu AM, Cionac Florescu S, Anastase DM, et al. Is there any analgesic benefit from preoperative vs. postoperative administration of etoricoxib in total knee arthroplasty under spinal anaesthesia?: A randomised double-blind placebo-controlled trial. Eur J Anaesthesiol. 2016;33(11):840-845.
31. Falci SGM, Lima TC, Martins CC, et al. Preemptive effect of dexamethasone in third-molar surgery: a meta-analysis. Anesth Prog. 2017;64(3):136-143.
32. Alcântara CE, Falci SG, Oliveira-Ferreira F, et al. Pre-emptive effect of dexamethasone and methylprednisolone on pain, swelling, and trismus after third molar surgery: a split-mouth randomized triple-blind clinical trial. Int J Oral Maxillofac Surg. 2014;43(1):93-98.
33. Donaldson M, Goodchild JG. Lidocaine turns 70: the evolution of dental local anesthesia. Gen Dent. 2018;66(3):6-9.
34. Chen Q, Chen J, Hu B, et al. Submucosal injection of dexamethasone reduces postoperative discomfort after third-molar extraction: a systematic review and meta-analysis. J Am Dent Assoc. 2017;148(2):81-91.
35. Dan AE, Thygesen TH, Pinholt EM. Corticosteroid administration in oral and orthognathic surgery: a systematic review of the literature and meta-analysis. J Oral Maxillofac Surg. 2010;68(9):2207-2220.
36. Dwyer Schull P. Dexamethasone. In: Johnson CA, Pancotti R, eds. Nurse's Drug Handbook. 7th ed. Plano, TX: McGraw-Hill Company, Inc; 2013:351.